- Calcium & bone metabolism
- Age-Dependent Association of Height Loss with Incident Fracture Risk in Postmenopausal Korean Women
-
Chaewon Lee, Hye-Sun Park, Yumie Rhee, Namki Hong
-
Endocrinol Metab. 2023;38(6):669-678. Published online September 1, 2023
-
DOI: https://doi.org/10.3803/EnM.2023.1734
-
-
 Abstract Abstract
 PDF PDF Supplementary Material Supplementary Material PubReader PubReader  ePub ePub
- Background
Height loss is a simple clinical measure associated with increased fracture risk. However, limited data exists on the association between height loss and fracture risk in postmenopausal Korean women. It is unknown whether this association varies with age.
Methods
Data on height loss over a 6-year period were collected from a community-based longitudinal follow-up cohort (Ansung cohort of the Korean Genome and Epidemiology Study). Incident fractures were defined based on self-reported fractures after excluding those due to severe trauma or toes/fingers. The association between incident fractures and height loss was investigated using a Cox proportional hazards model.
Results
During a median follow-up of 10 years after the second visit, 259/1,806 participants (median age, 64 years) experienced incident fractures. Overall, a 1 standard deviation (SD) decrease in height (1.6 cm/median 5.8 years) was associated with 9% increased risk of fracture (hazard ratio [HR], 1.09; P=0.037), which lost statistical significance after adjustment for covariates. When stratified into age groups (50–59, 60–69, 70 years or older), a 1 SD decrease in height remained a robust predictor of fracture in the 50 to 59 years age group after adjusting for covariates (adjusted hazard ratio [aHR], 1.52; P=0.003), whereas height loss was not an independent predictor of fracture in the 60 to 69 (aHR, 1.06; P=0.333) or the 70 years or older age groups (aHR, 1.05; P=0.700; P for interaction <0.05, for all).
Conclusion
Height loss during the previous 6 years was associated with an increased 10-year fracture risk in postmenopausal women in their 50s.
- Calcium & Bone Metabolism
- Update on Preoperative Parathyroid Localization in Primary Hyperparathyroidism
-
Hye-Sun Park, Namki Hong, Jong Ju Jeong, Mijin Yun, Yumie Rhee
-
Endocrinol Metab. 2022;37(5):744-755. Published online October 25, 2022
-
DOI: https://doi.org/10.3803/EnM.2022.1589
-
-
4,141
View
-
358
Download
-
5
Web of Science
-
6
Crossref
-
 Abstract Abstract
 PDF PDF PubReader PubReader  ePub ePub
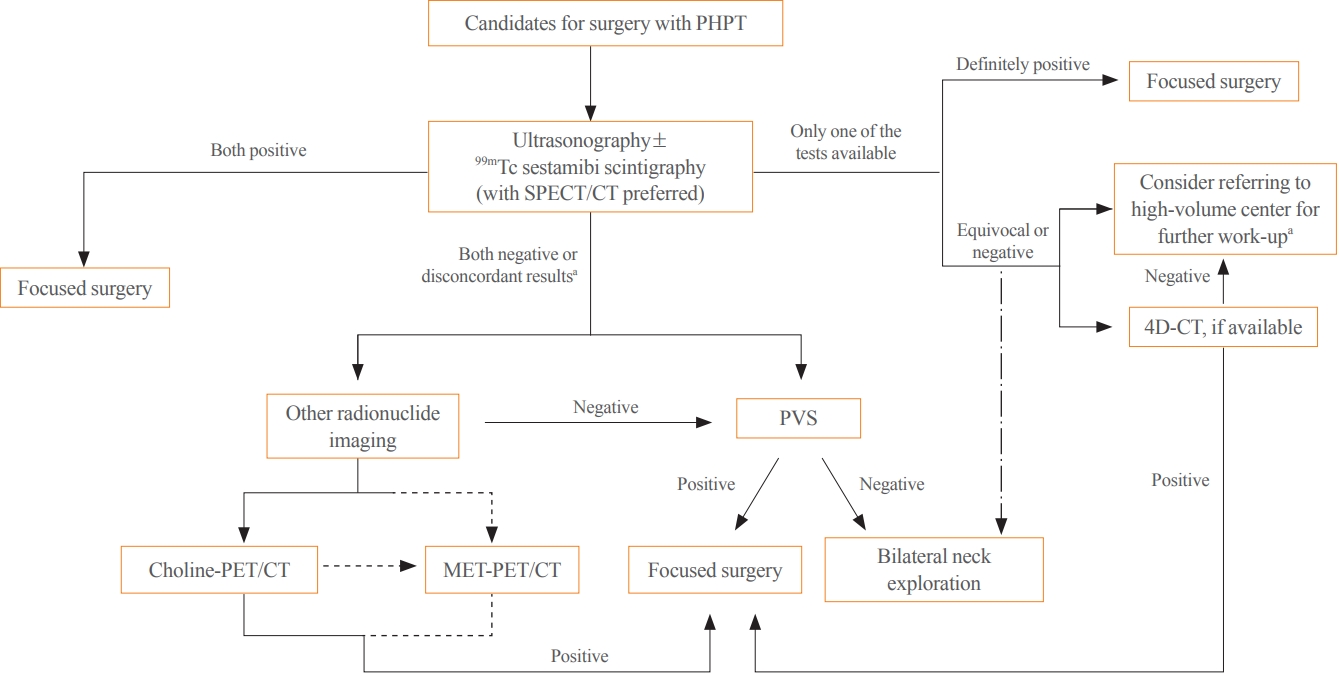
- Parathyroidectomy is the treatment of choice for primary hyperparathyroidism when the clinical criteria are met. Although bilateral neck exploration is traditionally the standard method for surgery, minimally invasive parathyroidectomy (MIP), or focused parathyroidectomy, has been widely accepted with comparable curative outcomes. For successful MIP, accurate preoperative localization of parathyroid lesions is essential. However, no consensus exists on the optimal approach for localization. Currently, ultrasonography and technetium-99m-sestamibi–single photon emission computed tomography/computed tomography are widely accepted in most cases. However, exact localization cannot always be achieved, especially in cases with multiglandular disease, ectopic glands, recurrent disease, and normocalcemic primary hyperparathyroidism. Therefore, new modalities for preoperative localization have been developed and evaluated. Positron emission tomography/computed tomography and parathyroid venous sampling have demonstrated improvements in sensitivity and accuracy. Both anatomical and functional information can be obtained by combining these methods. As each approach has its advantages and disadvantages, the localization study should be deliberately chosen based on each patient’s clinical profile, costs, radiation exposure, and the availability of experienced experts. In this review, we summarize various methods for the localization of hyperfunctioning parathyroid tissues in primary hyperparathyroidism.
-
Citations
Citations to this article as recorded by  - Expression of the Calcium-Sensing Receptor on Normal and Abnormal Parathyroid and Thyroid Tissue
Anne L. Worth, Mesrop Ayrapetyan, Susan J. Maygarden, Zibo Li, Zhanhong Wu, Chris B. Agala, Lawrence T. Kim
Journal of Surgical Research.2024; 293: 618. CrossRef - Use of [18F]fluorocholine PET/CT in the detection of primary hyperparathyroidism in paediatrics: a case report
Helena Martínez Sánchez, Francisca Moreno Macián, Sara León Cariñena, Carmen de Mingo Alemany, Lidia Blasco González, Raquel Sánchez Vañó
Journal of Pediatric Endocrinology and Metabolism.2024;[Epub] CrossRef - A Rare Case of Hyperfunctioning Lipoadenoma Presenting as a Cystic Pararthyroid Lesion
Jinyoung Kim, Ohjoon Kwon, Tae-Jung Kim, So Lyung Jung, Eun Ji Han, Ki-Ho Song
Journal of Bone Metabolism.2023; 30(2): 201. CrossRef - Role of 18F-Fluorocholine Positron Emission Tomography (PET)/Computed Tomography (CT) in Diagnosis of Elusive Parathyroid Adenoma
Janan R Badier, Pokhraj P Suthar, Jagadeesh S Singh, Miral D Jhaveri
Cureus.2023;[Epub] CrossRef - Pitfalls of DualTracer 99m-Technetium (Tc) Pertechnetate and Sestamibi Scintigraphy before Parathyroidectomy: Between Primary-Hyperparathyroidism-Associated Parathyroid Tumour and Ectopic Thyroid Tissue
Mara Carsote, Mihaela Stanciu, Florina Ligia Popa, Oana-Claudia Sima, Eugenia Petrova, Anca-Pati Cucu, Claudiu Nistor
Medicina.2023; 60(1): 15. CrossRef - Diagnostic Performance of Magnetic Resonance Imaging for Parathyroid Localization of Primary Hyperparathyroidism: A Systematic Review
Max H. M. C. Scheepers, Zaid Al-Difaie, Lloyd Brandts, Andrea Peeters, Bjorn Winkens, Mahdi Al-Taher, Sanne M. E. Engelen, Tim Lubbers, Bas Havekes, Nicole D. Bouvy, Alida A. Postma
Diagnostics.2023; 14(1): 25. CrossRef
- Diabetes, Obesity and Metabolism
- Association of Shift Work with Normal-Weight Obesity in Community-Dwelling Adults
-
Chul Woo Ahn, Sungjae Shin, Seunghyun Lee, Hye-Sun Park, Namki Hong, Yumie Rhee
-
Endocrinol Metab. 2022;37(5):781-790. Published online October 25, 2022
-
DOI: https://doi.org/10.3803/EnM.2022.1532
-
-
3,359
View
-
190
Download
-
3
Web of Science
-
3
Crossref
-
 Abstract Abstract
 PDF PDF Supplementary Material Supplementary Material PubReader PubReader  ePub ePub
- Background
Shift work is associated with obesity and metabolic syndrome. However, this association in the normal-weight population remains unclear. This study aimed to investigate whether shift work is associated with normal-weight obesity (NWO).
Methods
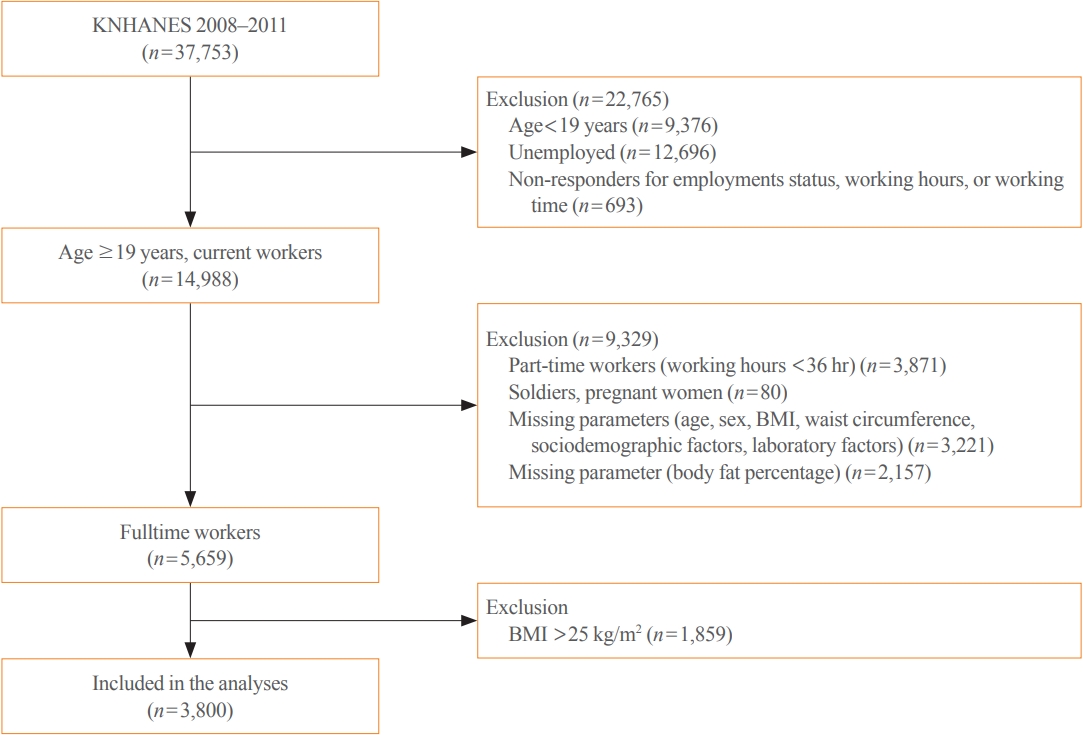
From the nationally representative Korea National Health and Nutrition Examination Survey (KNHANES) dataset (2008 to 2011), 3,800 full-time workers aged ≥19 years with a body mass index (BMI) ≤25 kg/m2 were analysed. We defined NWO as BMI ≤25 kg/m2 and body fat percentage ≥25% in men and ≥37% in women. Working patterns were classified into “daytime,” “other than daytime,” and “shift.” Multivariable logistic regression analysis was performed to evaluate the relationship between shift work and NWO.
Results
Shift work was associated with higher odds of NWO than daytime work (adjusted odds ratio [aOR], 1.47; 95% confidence interval [CI], 1.04 to 2.09) and night/evening work (aOR, 1.87; 95% CI, 1.11 to 3.14) after adjustment for type of work, working hours, age, sex, BMI, 25-hydroxyvitamin D levels, homeostatic model assessment for insulin resistance, and other sociodemographic factors. In subgroup analyses, the association between shift work and NWO was more robust in those aged ≥60 years and those working ≥56 hours/week.
Conclusion
Shift work was associated with NWO in community-dwelling Korean adults, independent of age, sex, BMI, and other covariates.
-
Citations
Citations to this article as recorded by  - Impaired Melatonin Secretion, Oxidative Stress and Metabolic Syndrome in Night Shift Work
Sorina Hohor, Cristina Mandanach, Andreea Maftei, Corina Aurelia Zugravu, Marina Ruxandra Oțelea
Antioxidants.2023; 12(4): 959. CrossRef - Normal-Weight Obesity and Metabolic Syndrome in Korean Adults: A Population-Based Cross-Sectional Study
Jeonghyeon Kim, Seamon Kang, Hyunsik Kang
Healthcare.2023; 11(16): 2303. CrossRef - You Can’t Avoid Shift Work? Then Focus on Body Fat Rather than Weight
Eun Kyung Lee
Endocrinology and Metabolism.2022; 37(5): 756. CrossRef
- Clinical Study
- Effects of Single Vitamin D3 Injection (200,000 Units) on Serum Fibroblast Growth Factor 23 and Sclerostin Levels in Subjects with Vitamin D Deficiency
-
Dongdong Zhang, Da Hea Seo, Han Seok Choi, Hye-Sun Park, Yoon-Sok Chung, Sung-Kil Lim
-
Endocrinol Metab. 2017;32(4):451-459. Published online December 14, 2017
-
DOI: https://doi.org/10.3803/EnM.2017.32.4.451
-
-
4,586
View
-
50
Download
-
8
Web of Science
-
10
Crossref
-
 Abstract Abstract
 PDF PDF PubReader PubReader
- Background
Vitamin D deficiency remains common in all age groups and affects skeletal and non-skeletal health. Fibroblast growth factor 23 is a bone-derived hormone that regulates phosphate and 1,25-dihydroxyvitamin D homeostasis as a counter regulatory factor. 1,25-Dihydroxyvitamin D stimulates fibroblast growth factor 23 synthesis in bone, while fibroblast growth factor 23 suppresses 1,25-dihydroxyvitamin D production in the kidney. The aim of this study was to evaluate the effects of vitamin D3 intramuscular injection therapy on serum fibroblast growth factor 23 concentrations, and several other parameters associated with bone metabolism such as sclerostin, dickkopf-1, and parathyroid hormone. MethodsA total of 34 subjects with vitamin D deficiency (defined by serum 25-hydroxyvitamin D levels below 20 ng/mL) were randomly assigned to either the vitamin D injection group (200,000 units) or placebo treatment group. Serum calcium, phosphate, urine calcium/creatinine, serum 25-hydroxyvitamin D, fibroblast growth factor 23, sclerostin, parathyroid hormone, and dickkopf-1 levels were serially measured after treatment. ResultsComparing the vitamin D injection group with the placebo group, no significant changes were observed in serum fibroblast growth factor 23, parathyroid hormone, or dickkopf-1 levels. Serum sclerostin concentrations transiently increased at week 4 in the vitamin D group. However, these elevated levels declined later and there were no statistically significant differences as compared with baseline levels. ConclusionSerum fibroblast factor 23, sclerostin, parathyroid hormone, and dickkopf-1 levels were not affected significantly by single intramuscular injection of vitamin D3.
-
Citations
Citations to this article as recorded by  - Effect of vitamin D supplementation on circulating fibroblast growth factor-23 concentration in adults with prediabetes
Lisa Ceglia, Anastassios G. Pittas, Bess Dawson-Hughes
Aging Clinical and Experimental Research.2023; 35(3): 525. CrossRef - Fibroblast Growth Factor 23 in COVID-19: An Observational Study
Athena Myrou, Theodoros Aslanidis, Keli Makedou, Athanasios Mitsianis, Aikaterini Thisiadou, Paraskevi Karalazou, Georgios Chatzopoulos, Anastasios Papadopoulos, Antonios Kalis, Dimitrios Giagkoulis, Fotios Lezgidis, Christos Savopoulos
Cureus.2023;[Epub] CrossRef - The effect of vitamin D supplementation on serum levels of fibroblast growth factor- 23: A systematic review and meta-analysis of randomized controlled trials
Fatemeh Meshkini, Sepideh Soltani, Cain C.T. Clark, Vivian Tam, David Meyre, Omid Toupchian, Sahar Saraf-Bank, Shima Abdollahi
The Journal of Steroid Biochemistry and Molecular Biology.2022; 215: 106012. CrossRef - Serum sclerostin levels in osteoporotic fracture patients
Erwin A. Gorter, Casper R. Reinders, Pieta Krijnen, Natasha M. Appelman-Dijkstra, Inger B. Schipper
European Journal of Trauma and Emergency Surgery.2022; 48(6): 4857. CrossRef - Clinical Utility of Preoperative Vitamin D3 Injection for Preventing Transient Hypocalcemia after Total Thyroidectomy
Kwangsoon Kim, Cho Rok Lee, Sang-Wook Kang, Jandee Lee, Jong Ju Jeong, Kee-Hyun Nam, Woong Youn Chung, Claudio Casella
International Journal of Endocrinology.2021; 2021: 1. CrossRef - The effect of vitamin D supplementation on fibroblast growth factor‐23 in patients with chronic kidney disease: A systematic review and meta‐analysis
Elmira Karimi, Sama Bitarafan, Seyed Mohammad Mousavi, Nikan Zargarzadeh, Pari Mokhtari, Jessie Hawkins, Alipasha Meysamie, Fariba Koohdani
Phytotherapy Research.2021; 35(10): 5339. CrossRef - Pharmacodynamics of Oral Cholecalciferol in Healthy Individuals with Vitamin D Deficiency: A Randomized Open-Label Study
Angelo Fassio, Davide Gatti, Maurizio Rossini, Camilla Benini, Elena Fracassi, Eugenia Bertoldo, Ombretta Viapiana, Stefano Milleri, Matteo Gatti, Giovanni Adami
Nutrients.2021; 13(7): 2293. CrossRef - Vitamin D Deficiency at Mid-Pregnancy Is Associated with a Higher Risk of Postpartum Glucose Intolerance in Women with Gestational Diabetes Mellitus
Kyung-Soo Kim, Seok Won Park, Yong-Wook Cho, Soo-Kyung Kim
Endocrinology and Metabolism.2020; 35(1): 97. CrossRef - Effects of vitamin D supplementation on bone turnover markers and other bone-related substances in subjects with vitamin D deficiency
Rolf Jorde, Astrid Kamilla Stunes, Julia Kubiak, Ragnar Joakimsen, Guri Grimnes, Per Medbøe Thorsby, Unni Syversen
Bone.2019; 124: 7. CrossRef - Vitamin D Enhances the Efficacy of Topical Artificial Tears in Patients With Dry Eye Disease
Jin Sun Hwang, Yoon Pyo Lee, Young Joo Shin
Cornea.2019; 38(3): 304. CrossRef
- Site-Specific Difference of Bone Geometry Indices in Hypoparathyroid Patients
-
Hye-Sun Park, Da Hea Seo, Yumie Rhee, Sung-Kil Lim
-
Endocrinol Metab. 2017;32(1):68-76. Published online February 6, 2017
-
DOI: https://doi.org/10.3803/EnM.2017.32.1.68
-
-
3,332
View
-
32
Download
-
3
Web of Science
-
4
Crossref
-
 Abstract Abstract
 PDF PDF PubReader PubReader
- Background
Hypoparathyroid patients often have a higher bone mineral density (BMD) than the general population. However, an increase in BMD does not necessarily correlate with a solid bone microstructure. This study aimed to evaluate the bone microstructure of hypoparathyroid patients by using hip structure analysis (HSA). MethodsNinety-five hypoparathyroid patients >20 years old were enrolled and 31 of them had eligible data for analyzing bone geometry parameters using HSA. And among the control data, we extracted sex-, age-, and body mass index-matched three control subjects to each patient. The BMD data were reviewed retrospectively and the bone geometry parameters of the patients were analyzed by HSA. ResultsThe mean Z-scores of hypoparathyroid patients at the lumbar spine, femoral neck, and total hip were above zero (0.63±1.17, 0.48±1.13, and 0.62±1.10, respectively). The differences in bone geometric parameters were site specific. At the femoral neck and intertrochanter, the cross-sectional area (CSA) and cortical thickness (C.th) were higher, whereas the buckling ratio (BR) was lower than in controls. However, those trends were opposite at the femoral shaft; that is, the CSA and C.th were low and the BR was high. ConclusionOur study shows the site-specific effects of hypoparathyroidism on the bone. Differences in bone components, marrow composition, or modeling based bone formation may explain these findings. However, further studies are warranted to investigate the mechanism, and its relation to fracture risk.
-
Citations
Citations to this article as recorded by  - Vertebral fractures, trabecular bone score and their determinants in chronic hypoparathyroidism
S. Saha, V. Mannar, D. Kandasamy, V. Sreenivas, R. Goswami
Journal of Endocrinological Investigation.2022; 45(9): 1777. CrossRef - Epidemiology and Financial Burden of Adult Chronic Hypoparathyroidism
Sigridur Bjornsdottir, Steven Ing, Deborah M Mitchell, Tanja Sikjaer, Line Underbjerg, Zaki Hassan-Smith, Jad Sfeir, Neil J Gittoes, Bart L Clarke L
Journal of Bone and Mineral Research.2020; 37(12): 2602. CrossRef - Effect of Endogenous Parathyroid Hormone on Bone Geometry and Skeletal Microarchitecture
A Ram Hong, Ji Hyun Lee, Jung Hee Kim, Sang Wan Kim, Chan Soo Shin
Calcified Tissue International.2019; 104(4): 382. CrossRef - Bone responses to chronic treatment of adult hypoparathyroid patients with PTH peptides
Sofie Malmstroem, Lars Rejnmark, Dolores M. Shoback
Current Opinion in Endocrine and Metabolic Research.2018; 3: 51. CrossRef
- Adrenal gland
- Untreated Congenital Adrenal Hyperplasia with 17-α Hydroxylase/17,20-Lyase Deficiency Presenting as Massive Adrenocortical Tumor
-
Su Jin Lee, Je Eun Song, Sena Hwang, Ji-Yeon Lee, Hye-Sun Park, Seunghee Han, Yumie Rhee
-
Endocrinol Metab. 2015;30(3):408-413. Published online August 4, 2015
-
DOI: https://doi.org/10.3803/EnM.2015.30.3.408
-
-
4,264
View
-
47
Download
-
3
Web of Science
-
4
Crossref
-
 Abstract Abstract
 PDF PDF PubReader PubReader
Congenital adrenal hyperplasia (CAH) with 17α-hydroxylase/17,20-lyase deficiency is usually characterized by hypertension and primary amenorrhea, sexual infantilism in women, and pseudohermaphroditism in men. hypertension, and sexual infantilism in women and pseudohermaphroditism in men. In rare cases, a huge adrenal gland tumor can present as a clinical manifestation in untreated CAH. Adrenal cortical adenoma is an even more rare phenotype in CAH with 17α-hydroxylase/17,20-lyase deficiency. A 36-year-old female presented with hypertension and abdominal pain caused by a huge adrenal mass. Due to mass size and symptoms, left adrenalectomy was performed. After adrenalectomy, blood pressure remained high. Based on hormonal and genetic evaluation, the patient was diagnosed as CAH with 17α-hydroxylase/17,20-lyase deficiency. The possibility of a tumorous change in the adrenal gland due to untreated CAH should be considered. It is important that untreated CAH not be misdiagnosed as primary adrenal tumor as these conditions require different treatments. Adequate suppression of adrenocorticotropic hormone (ACTH) in CAH is also important to treat and to prevent the tumorous changes in the adrenal gland. Herein, we report a case of untreated CAH with 17α-hydroxylase/17,20-lyase deficiency presenting with large adrenal cortical adenoma and discuss the progression of adrenal gland hyperplasia due to inappropriate suppression of ACTH secretion. -
Citations
Citations to this article as recorded by  - Congenital adrenal hyperplasia disorder due to 17 α-hydroxylase deficiency: a case report
Yunling Tian, Lijie Hou, Shulan Xiang, Xuguang Tian, Jinhui Xu
Gynecological Endocrinology.2023;[Epub] CrossRef - Landscape of Adrenal Tumours in Patients with Congenital Adrenal Hyperplasia
Mara Carsote, Ana-Maria Gheorghe, Claudiu Nistor, Alexandra-Ioana Trandafir, Oana-Claudia Sima, Anca-Pati Cucu, Adrian Ciuche, Eugenia Petrova, Adina Ghemigian
Biomedicines.2023; 11(11): 3081. CrossRef - 17α-Hydroxylase/17,20-Lyase Deficiency in 46,XY: Our Experience and Review of Literature
Madhur Maheshwari, Sneha Arya, Anurag Ranjan Lila, Vijaya Sarathi, Rohit Barnabas, Khushnandan Rai, Vishwambhar Vishnu Bhandare, Saba Samad Memon, Manjiri Pramod Karlekar, Virendra Patil, Nalini S Shah, Ambarish Kunwar, Tushar Bandgar
Journal of the Endocrine Society.2022;[Epub] CrossRef - 17α-hydroxylase Deficiency Mimicking Hyperaldosteronism by Aldosterone-producing Adrenal Adenoma
Yun Kyung Cho, Hyeseon Oh, Sun-myoung Kang, Sujong An, Jin-Young Huh, Ji-Hyang Lee, Woo Je Lee
The Korean Journal of Medicine.2016; 91(2): 191. CrossRef
|